This month we met Anna Corti, 29, now a researcher at B3Lab. Her research, part of the AI-CORPS project, aims to create a predictive model of atherosclerotic plaque rupture in coronary arteries, which will offer great benefits to both patients and healthcare systems.

Always ‘loyal’ to Politecnico di Milano, her academic career began here with her Laurea Triennale and Laurea Magistrale in Biomedical Engineering and continued with her three-year PhD.
Precisely for the innovative aspects of her doctoral thesis, she received several awards, including the Best Doctoral Thesis Award in Biomechanics from the European Society of Biomechanics (ESB), the VPHi Best Thesis Award in In Silico Medicine from the Virtual Physiological Human Institute (VPHi) and the Best Doctoral Thesis Award in Theoretical and Applied Biomechanics from the Biomechanics Group of the Italian Association of Theoretical and Applied Mechanics (GBMA-AIMETA).
Welcome, Anna. Where did you carry out your research here at Politecnico?
For my Laurea Magistrale thesis, I started working at LaBS CompBiomech, the Computational Biomechanics Laboratory of the ‘Giulio Natta’ Department of Chemistry, Materials and Chemical Engineering. Also for that thesis, I did a research period abroad at the Houston Methodist Hospital. There I became more and more passionate about research. The next step was to choose to do a PhD and do it at LaBS, under the supervision of Professors Francesco Migliavacca, José Felix Rodriguez Matas and Claudio Chiastra. I completed this course of studies last July.
After this milestone, which road did you take?
I continued my research at LaBS, but started a collaboration with B3Lab, a biosignals, bioimaging and bioinformatics laboratory that focuses on the development of predictive artificial intelligence methods based on the analysis of biomedical signals, images and multi-omics data with the aim of contributing to the improvement of therapies in the context of personalised and precision medicine. In the last year, I officially moved to this lab as a post-doc, and then transferred to the Department of Electronics, Information and Bioengineering.
What did you do during your PhD?
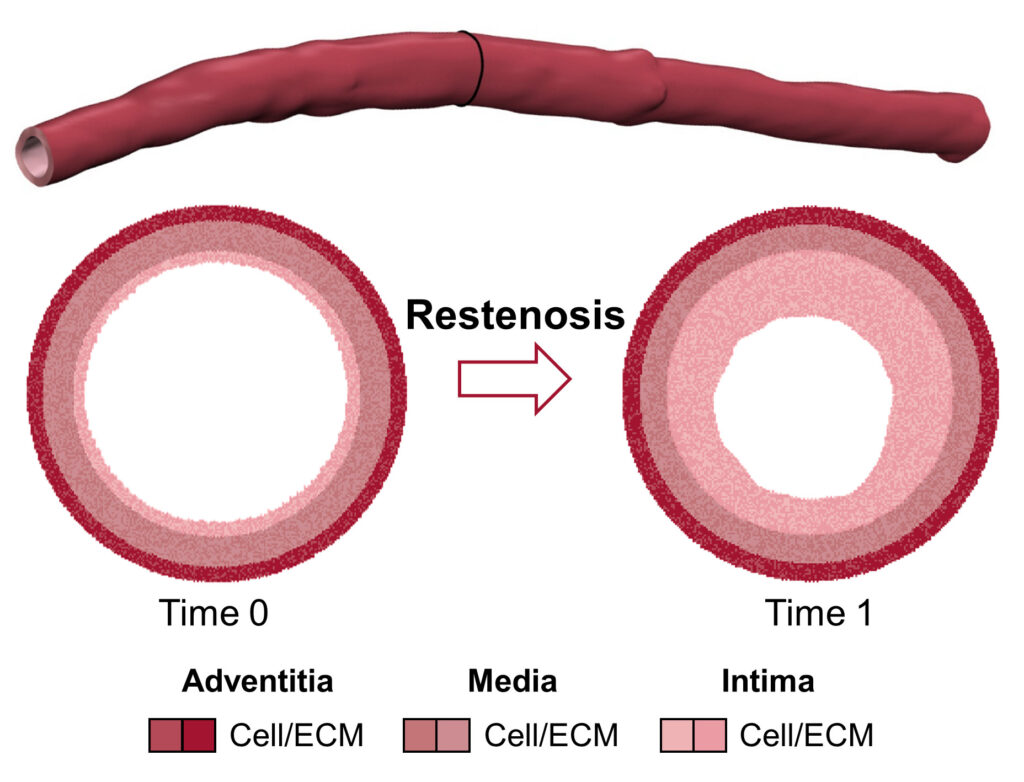
During my PhD, I specialised in in silico (ie computational) multiscale modelling to study vascular remodelling processesduring the development of atherosclerosis and restenosis.
My work was part of the project ‘TIME – From TIssue to Molecular mechanisms of restenosis after peripheral Endovascular interventions’, funded by Fondazione Cariplo and coordinated by Professor Claudio Chiastra. Our LaBS team was joined by other international partners: University of Florida, Houston Methodist Hospital and Politecnico di Torino.
In particular, I developed a patient-specific model based on several computational methods (CFD simulations and agent-based model) to study the effects of local haemodynamics and the level of systemic inflammation on the outcome of endovascular procedures, and thus on restenosis, using the emerging ‘systems biology’ approach. The aim was to understand the processes underlying restenosis, which is currently one of the main causes of failure in endovascular procedures.
What procedures are we talking about?
These are minimally invasive procedures that serve to restore blood flow in the arteries, which was previously reduced due to the formation of atherosclerotic plaques that lead to their narrowing. The formation of these plaques is due to atherosclerosis, a vascular disease commonly associated with ageing and favoured by various factors such as high cholesterol levels, blood pressure, the presence of diabetes and bad lifestyles: in terms of diet, physical exercise, substances such as alcohol and smoking.
One of the most frequently used procedures in these cases to keep the vessel patent (open) is the implantation of a stent, a kind of small metal-mesh scaffold, which is delivered to the vessel through a catheter. The stent can either self-expand or be expanded by inflating a balloon, which is then deflated and extracted.
The problem is that in some cases the artery reacts to the intervention with an inflammatory process that causes an abnormal growth of the artery wall itself, returning the vessel to a condition of partial or total occlusion (as it was previously). In these cases we speak of restenosis: it is a failure of the procedure, which we are analysing via this research.
What has this research meant to you?
It demonstrated how mathematical models and computational simulations are high-potential tools, which, by providing a virtual environment to test clinical hypotheses, can contribute to the understanding of the complex processes underlying pathology and guide the development of more effective therapies aimed at improving post-surgical outcome.
An important innovation of this study involved the integration of patient-specific gene expression data with a model replicating cellular dynamics, which laid the foundation towards the development of a multiscale ‘in silico’ medicine approach.
Thus, it is not by focusing only on a single factor, but rather on the integration of many of these – from the molecular to the cellular and tissue scale – that we can understand the complex biological mechanisms at play in the response to a stent implant.
How did you move to the Department of Electronics, Information and Bioengineering?
The research carried out during my PhD was aimed at understanding the multiscale and multifactorial mechanical-biological mechanisms underlying these vascular processes and their interaction. We succeeded in developing a framework that, by integrating inflammatory (via a gene expression network) and haemodynamic effects on cellular dynamics, was able to predict restenosis in patient-specific stented arteries up to one year after surgery. By validating the framework, we thus confirmed the model’s assumptions based on the integration of these factors as a key element in the development of restenosis. The study was therefore mainly based on a computational biomechanicsapproach. At that point, I was convinced that integration with artificial intelligence methodscould help us go further in the development of a predictive model.
I got in touch with professors Luca Mainardi and Valentina Corino from B3Lab in order to supplement my biomechanical background with AI knowledge.
What project are you now working on at DEIB?
The project is called AI-CORPS – ‘Trustworthy, integrated Artificial Intelligence tools for predicting high-risk CORonary PlaqueS’ and is funded by Fondazione Regionale per la Ricerca Biomedica. We are working on this project together with the Centro Cardiologico Monzino, the Policlinico San Matteo and the San Raffaele Hospital.
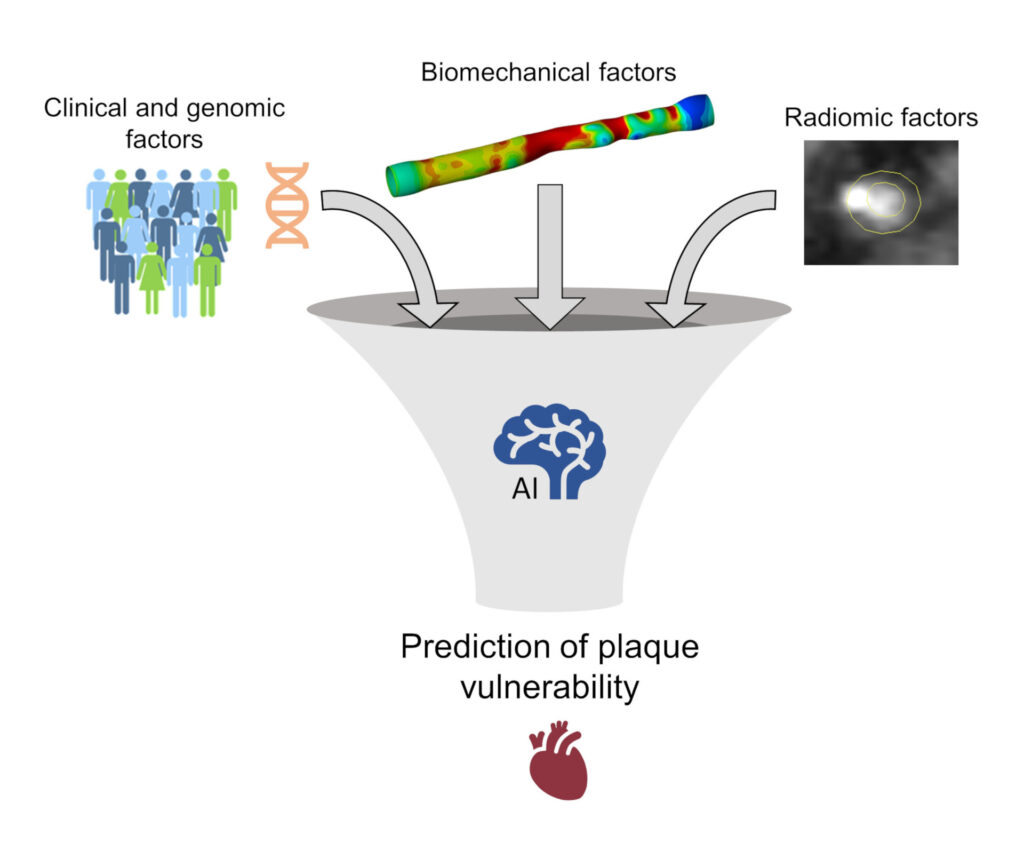
The aim is to develop a predictive model of atherosclerotic plaque rupture in coronary arteriesby integrating clinical (such as the presence of diabetes and other comorbidities), genomic, biomechanical and radiomic (extracted from radiological images) biomarkers.
The underlying hypothesis is that an approach integrating factors of different origins allows better identification of patients at cardiovascular risk and thus advances in terms of diagnosis and treatment, contributing to the development of personalised treatments in the field of precision medicine.
What do you prize in this approach?
This project fascinates me a lot, because it integrates biomechanical factors such as fluid dynamics and wall stress (obtained through computational simulations) with clinical, genomic and image analysis data. All this is supplemented with artificial intelligence methods.
The underlying element is multidisciplinarity, which is one of the aspects of biomedical research that stimulates me the most. These complex issues require strong integration between different worlds – that of doctors, chemists, biologists, engineers – using different languages.
Working together is the only way there is to find solutions after understanding the underlying phenomena.
As for the tool you all are designing, what will their benefits be?
With this new research, the focus is on providing a tool for clinicians to help them assess whether one plaque is more at risk than another.
There would be several benefits: on the one hand, it would be possible to identify patients at risk of an acute cardiovascular event (such as a heart attack), in order to then intervene more quickly and with a greater likelihood of a positive outcome for the patient; on the other hand, overtreatment of patients who are not really at risk could be avoided, so as to reduce unnecessary interventions. Hence, better prevention, diagnosis and management of the disease, with reduced risks for the patient and reduced costs and burden on the healthcare system.
Some clinical methods for risk assessment already exist today, but their accuracy is still suboptimal. The development of these AI-based predictive models will be an important decision-making support to identify patients more at risk than others.
Do you already have clear plans for the future?
Next year I am here at DEIB, I will stay on this project.
But I have to be honest: I have considered pursuing an academic career… and I would like to. I am a curious, creative person and I believe that curiosity lies at the heart of the world of research. Knowledge can only increase, it does not stop. And for this to happen, collaboration is crucial.
I have also had teaching experience with tutorials. This is another aspect that I am passionate about. Because stimulating students and arousing their interest is very rewarding. Training future engineers must be a beautiful mission.
As a university career combines these two aspects, this is what I hope to continue doing.
How did you choose Politecnico di Milano and how are you coping?
From a scientific point of view, it is a centre of reference and prestige, recognised worldwide, even in the United States, where I did my thesis.
Humanly speaking, I always got along well, both at LaBS and B3Lab. It is a place where people like to work.

How did it all start with your love of engineering?
Actually, I attended a modern language high school. But I think that at 13 you still have to find out what you like. I had a maths and physics teacher who motivated me a lot when I decided to choose this path.
I was hesitating at first: I was the only one in the whole institute to undertake an engineering science programme. Of course, it was more tiring in the first year, but with willpower and commitment I learnt the basics and caught up. Today I can say that it is a feasible course of study even for those who did not attend a scientific high school if there is passion.
Is being a woman and doing engineering still considered something uncommon? How did you experience it?
In fact, biomedicine is one of those engineering courses where the situation is quite balanced, as far as the gender of the enrolled students is concerned. There is also a strong female component both at LaBS and at B3Lab.
Anyway, I believe that this situation of inequality is becoming narrower and narrower. During these years, I have attended congresses, conferences, both European and international, and many women have been awarded for the quality of their research work. I have never witnessed any discrimination in this field.
For those who are still undecided, would you recommend enrolling in engineering?
Of course I do, regardless of one’s gender and course of study. The only thing that cannot be lacking is motivation, as a lot of it is needed. Everyone has their own path, we are all different. But the moment you realise what yours is, you go straight to the goal if you are inspired.
As far as biomedicine is concerned, what motivates me a lot about research in this field is the direct impact we have on human lives. Perhaps what I am studying now is chronologically very far away from the time when it will have its application. But to know that developing methods that improve diagnostics, disease management and outcome will have a direct impact on all of us, on society, on modern medicine, is hugely inspiring!

